Is Dmg A Strong Field Ligand
A strong ligand or a strong field ligand is a ligand that can result in a higher crystal field splitting. This means, the binding of a strong field ligand causes a higher difference between the higher and lower energy level orbitals. Examples include CN – (cyanide ligands), NO 2– (nitro ligand) and CO (carbonyl ligands).
A spectrochemical series is a list of ligands ordered on ligand strength and a list of metal ions based on oxidation number, group and its identity. In crystal field theory, ligands modify the difference in energy between the d orbitals (Δ) called the ligand-field splitting parameter for ligands or the crystal-field splitting parameter, which is mainly reflected in differences in color of similar metal-ligand complexes.
Spectrochemical series of ligands[edit]
The spectrochemical series was first proposed in 1938 based on the results of absorption spectra of cobalt complexes.[1]
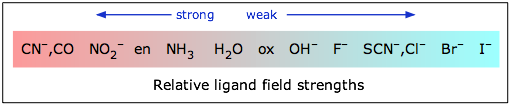
A partial spectrochemical series listing of ligands from small Δ to large Δ is given below. (For a table, see the ligand page.)
You do not add Dex mod to damage, however if you have a negative Str mod you do subtract from damage. If it is a composite longbow, it will have an amount of Str mod you can add to damage. Magic weapons also add to bow damage. Thrown weapons use dex mod for attack rolls and require you to add your Str mod to damage. Question Bows, Do you add str dex or nothing to damage? See title, please show me where it says this. With a strength bonus equal to or higher than the bow, you can add the bonus of the bow to damage. With a strength bonus lower than the bow, you cannot fire the bow or at least without a -4 bonus. As for where, I'm not certain. Nov 01, 2011 Thrown weapons or a composite strength bow lets you add in your Str mod for damage. I'll have to look up Dead eye, do you know what book it's in? I did see something in a dragon compilation that allowed you to add your dex modifier to damage, but it required a BAB of 12 or so. Bow Dexterity Damage Modifier 5th Edition. When firing a Longbow I know that you add your Dex Mod and Proficiency bonus to the attack roll but do you also add your Dex Mod to the damage roll? Save hide report. This thread is archived. New comments cannot be posted and votes cannot be cast. Do you add dex to bow dmg. UPDATE: the MM has a few other examples that are not consistent with 'Dex on damage', but in general, most are accurate. The answer below seems to back my.
I− < Br− < S2− < SCN− (S–bonded) < Cl− < N3− < F−< NCO− < OH− < C2O42− < O2−< H2O < acac− (acetylacetonate) < NCS− (N–bonded) < CH3CN < gly (glycine) < py (pyridine) < NH3 < en (ethylenediamine) < bipy (2,2'-bipyridine) < phen (1,10-phenanthroline) < NO2− < PPh3 < CN− < CO
Weak field ligand: H2O,F-,Cl-,OH-Strong field ligand: CO,CN-,NH3,PPh3
Ligands arranged on the left end of this spectrochemical series are generally regarded as weaker ligands and cannot cause forcible pairing of electrons within the 3d level, and thus form outer orbital octahedral complexes that are high spin. On the other hand, ligands lying at the right end are stronger ligands and form inner orbital octahedral complexes after forcible pairing of electrons within 3d level and hence are called low spin ligands.
However, keep in mind that 'the spectrochemical series is essentially backwards from what it should be for a reasonable prediction based on the assumptions of crystal field theory.'[2] This deviation from crystal field theory highlights the weakness of crystal field theory's assumption of purely ionic bonds between metal and ligand.
The order of the spectrochemical series can be derived from the understanding that ligands are frequently classified by their donor or acceptor abilities. Some, like NH3, are σ bond donors only, with no orbitals of appropriate symmetry for π bonding interactions. Bonding by these ligands to metals is relatively simple, using only the σ bonds to create relatively weak interactions. Another example of a σ bonding ligand would be ethylenediamine, however ethylenediamine has a stronger effect than ammonia, generating a larger ligand field split, Δ.
Ligands that have occupied p orbitals are potentially π donors. These types of ligands tend to donate these electrons to the metal along with the σ bonding electrons, exhibiting stronger metal-ligand interactions and an effective decrease of Δ. Most halide ligands as well as OH− are primary examples of π donor ligands.
When ligands have vacant π* and d orbitals of suitable energy, there is the possibility of pi backbonding, and the ligands may be π acceptors. This addition to the bonding scheme increases Δ. Ligands that do this very effectively include CN−, CO, and many others.[3]
Spectrochemical series of metals[edit]

The metal ions can also be arranged in order of increasing Δ, and this order is largely independent of the identity of the ligand.[4]
Mn2+ < Ni2+ < Co2+ < Fe2+ < V2+ < Fe3+ < Cr3+ < V3+ < Co3+
In general, it is not possible to say whether a given ligand will exert a strong field or a weak field on a given metal ion. However, when we consider the metal ion, the following two useful trends are observed:
Ligand Field Strength List
- Δ increases with increasing oxidation number, and
- Δ increases down a group.[4]
See also[edit]
Strong Field Ligand Example
References[edit]
- Zumdahl, Steven S. Chemical Principles Fifth Edition. Boston: Houghton Mifflin Company, 2005. Pages 550-551 and 957-964.
- D. F. Shriver and P. W. Atkins Inorganic Chemistry 3rd edition, Oxford University Press, 2001. Pages: 227-236.
- James E. Huheey, Ellen A. Keiter, and Richard L. Keiter Inorganic Chemistry: Principles of Structure and Reactivity 4th edition, HarperCollins College Publishers, 1993. Pages 405-408.
- ^R. Tsuchida (1938). 'Absorption Spectra of Co-ordination Compounds. I.'Bull. Chem. Soc. Jpn. 13 (5). doi:10.1246/bcsj.13.388.
- ^7th page of http://science.marshall.edu/castella/chm448/chap11.pdf
- ^Miessler, Gary; Tarr, Donald (2011). Inorganic Chemistry (4th ed.). Prentice Hall. pp. 395–396. ISBN978-0-13-612866-3.
- ^ abhttp://www.everyscience.com/Chemistry/Inorganic/Crystal_and_Ligand_Field_Theories/b.1013.php